Media Contacts:
Emily Makowski, 617-468-8970, [email protected]
Serena Bronda, 857-869-7828, [email protected]
Key Takeaways
- Researchers conducted a longitudinal analysis of HIV-1 reservoirs in people who have been on ART for approximately two decades.
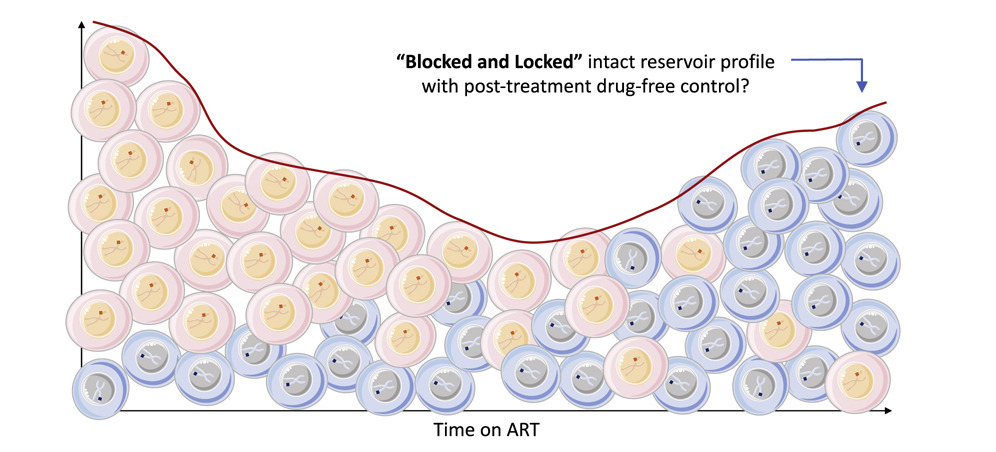
- The findings suggest that long-term ART causes the immune system to select for latent HIV reservoirs that are in integrated in “block and lock” positions in the human genome, where they cannot be easily reactivated and are less likely to produce infectious virus.
BOSTON – Antiretroviral therapy (ART) is highly effective in suppressing HIV and is recommended for all people who are HIV-positive. But although ART prevents new cells from being infected, it does not get rid of cells that are already infected. HIV infection creates a reservoir of infected immune system cells in the body that cannot be eliminated by antiretroviral therapy (ART), which is why there is currently no cure. Some HIV-positive individuals, however, termed “elite controllers,” do not have detectable viral loads of HIV even though they are not on ART. Researchers at Mass General Brigham’s founding members, Brigham and Women’s Hospital (BWH) and Massachusetts General Hospital (MGH); the Ragon Institute of Mass General, MIT, and Harvard; and colleagues have found that viral reservoirs in people who have been taking ART long-term have some similar characteristics to reservoirs in elite controllers. The results, published in Cell Host & Microbe today, suggest that continuous long-term ART can induce what the researchers call “deep latency,” where the virus is so silenced that some HIV-positive individuals may be able to avoid or delay developing symptoms in the event that treatment is interrupted.
Mathias Lichterfeld, MD, PhD, senior staff physician at Brigham and Women’s Hospital and Massachusetts General Hospital, professor of Medicine at Harvard Medical School, and member of the Ragon Institute of Mass General, MIT, and Harvard, and colleagues, previously demonstrated that genome-intact HIV DNA in elite controllers is integrated in highly selected parts of the human genome that repress HIV from being reactivated. They hypothesize that such a “block and lock” viral reservoir profile is the result of immune selection mechanisms. The immune system from elite controllers appears to preferentially eliminate viral genetic material located in areas of cells’ chromosomes that support viral transcriptional activity, while intact genetic material is “hidden” in areas where it is not likely to be transcribed and detected by the immune system. The researchers set out to discover whether this was also true for people who have been on ART long-term.
They evaluated peripheral blood mononuclear cells (PBMCs) from the viral reservoirs of eight participants who had been on ART for a median of 20 years using multiple viral sequencing assays: FLIP-Seq, MIP-Seq, PRIP-Seq, and quantitative viral outgrowth assays (qVOA). Just like in elite controllers, the participants on long-term ART also showed viral genomes in areas of the cells’ DNA that were not very transcriptionally active, while cells with viral genes integrated in genomic regions likely to be transcribed were eliminated over time.
In a previous study, the researchers looked at the effects of ART treatment interruption. The new study also looked at viral genetic material from participants in the previous study: two who had developed drug-free control of HIV after discontinuing ART, and three who rebounded and had high levels of the virus. Those who were able to maintain control of HIV without ART showed a bias toward having viral genetic material in areas that were less likely to be transcribed.
This research provides evidence that HIV reservoir cells are subject to immune selection mechanisms that, over many years of antiretroviral therapy, transform viral reservoirs. The findings suggest that after many years of taking ART, it may be possible for some people to discontinue ART without viral rebound; this idea is currently being tested in clinical trials. Further research is needed to clarify whether these selection mechanisms lead to clinically significant benefits.
“This study suggests that over very long periods of antiretroviral therapy, the immune system is able to corner viral reservoir cells, to an extent that only a small number of viruses integrated in highly repressive parts of the human genome can persist. We hypothesize that these persisting viruses are in “block and lock” positions and may have a more limited ability to drive viral rebound in case of treatment interruptions,” says Lichterfeld, the senior author of the study.
This research was supported in part by NIH grants AI117841, AI120008, AI152979, AI130005, DK120387, AI155233, AI169768, HL134539, AI116228, AI078799, DA047034, AI155171, AI150396,and amfAR (110181-69-RGCV), the Bill and Melinda Gates Foundation, (INV-002703) and amfAR ARCHE grant # 110393-72-RPRL, the DARE, ERASE, PAVE and BEAT-HIV Martin Delaney Collaboratories (UM1 AI164560, AI164562, AI164566, AI164570), the Delaney AIDS Research Enterprise (DARE; AI096109, AI127966) the amfAR Institute for HIV Cure Research (amfAR 109301), the Harvard University Center for AIDS Research (NIH P30 AI060354), the AIDS Clinical Trials Group (NIH/NIAID 2 UMAI069412-09), NIH NIAID: UM1AI068636, UM1AI106701, UM1AI068634, UM1AI069481, the Frederick National Laboratory for Cancer Research under Contract No. HHSN261200800001E, and the Intramural Research Program of the NIH, Frederick National Lab, Center for Cancer Research.
About the Ragon Institute of Mass General, MIT, and Harvard
The Ragon Institute of Mass General, MIT, and Harvard was established in 2009 with a gift from the Phillip T. and Susan M. Ragon Foundation, with a collaborative scientific mission among these institutions to harness the immune system to combat and cure human disease. Focusing on global infectious diseases, the Ragon Institute draws scientists, clinicians and engineers from diverse backgrounds and areas of expertise to study and understand the immune system with the goal of benefiting patients. For more information, visit www.ragoninstitute.org.
About Massachusetts General Hospital
Massachusetts General Hospital, founded in 1811, is the original and largest teaching hospital of Harvard Medical School. The Mass General Research Institute conducts the largest hospital-based research program in the nation, with annual research operations of more than $1 billion and comprises more than 9,500 researchers working across more than 30 institutes, centers and departments. In August 2021, Mass General was named #5 in the U.S. News & World Report list of “America’s Best Hospitals.” MGH is a founding member of the Mass General Brigham health care system.
About Brigham and Women’s Hospital
Brigham and Women’s Hospital is a founding member of Mass General Brigham and a teaching affiliate of Harvard Medical School. With nearly 1,000 inpatient beds, approximately 50,000 inpatient stays, and over 2.6 million outpatient encounters annually, clinicians across the Brigham provide compassionate, high-quality care in virtually every medical and surgical specialty to patients locally, regionally, nationally and around the world. An international leader in basic, clinical, and translational research, Brigham and Women’s Hospital has nearly 5,000 scientists, including physician-investigators, renowned biomedical researchers and faculty supported by nearly $750 million in funding. The Brigham’s medical preeminence and service to the community dates to 1832, with the opening of the Boston Lying In, one of the nation’s first maternity hospitals designed to care for women unable to afford in-home medical care. Its merger with the Free Hospital for Women resulted in the Boston Hospital for Women in 1966. In 1980, the Boston Hospital for Women, the Peter Bent Brigham Hospital and the Robert Breck Brigham Hospital officially merged to become Brigham and Women’s Hospital. With nearly 21,000 employees across the Brigham family – including the Brigham and Women’s Physicians Organization and Brigham and Women’s Faulkner Hospital – that rich history is the foundation for our commitment to providing superb care for some of the most complex cases, pursuing breakthroughs in biomedical research, training the next generation of health care providers, and serving the local and global community.

 Ragon Institute
Ragon Institute