Date: February 22, 2021 By:
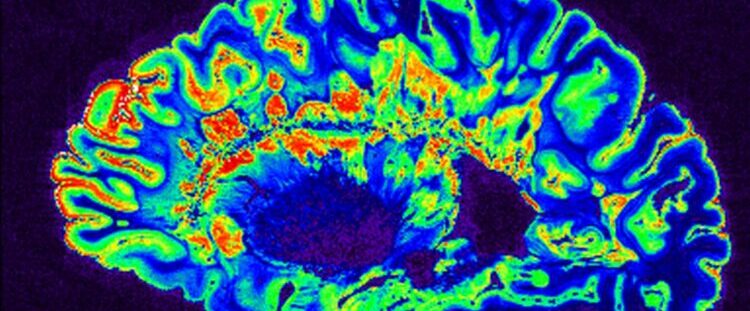
by: Rachel Leeson. Image credit: MRI Scan, courtesy of the NIH.
A stroke, like any traumatic event in the body, triggers an immune response and understanding this immune response is critical to improve patient outcomes and nominate new therapeutics. But immune responses in the brain are difficult to study in humans, because the brain is closed off from the rest of the body.
The MISTIE III clinical trial offered researchers like Alex Shalek, PhD, a core member of the Ragon Institute of Mass General, MIT, and Harvard, a unique opportunity to analyze this response through collecting clinical samples directly from patients who were in the acute recovery stage following a stroke, with the results recently published in Science Immunology. Dr. Lauren Sansing, MD, Academic Chief of the Division of Stroke and Vascular Neurology at Yale University, ran a sub-study of the trial called ICH-seq, in which the blood clots from the stroke patients were collected for cellular analysis. The trial itself used a flexible drain to treat patients who had suffered an intracerebral hemorrhage (ICH), in which there’s bleeding within the brain itself. The surgically implanted device allowed doctors to drain the hematoma over the five days following ICH. The fluid collected from the hematomas, and the immune cells inside, were used by researchers to gain rare insight into stroke recovery in real-time.
“This work led us to define the distinct responses of these immune cells in the hematoma during stroke recovery,” says Shalek, who is also an Institute Member of the Broad Institute of MIT and Harvard and a Core Member of the Institute for Medical Engineering and Science, an Associate Professor of Chemistry, and an Extramural Member of the Koch Institute for Cancer Research at MIT. “For the first time, our team was able to delineate the pathways and genes driving initial inflammation and subsequent increases in the expression of anti-inflammatory genes associated with resolution of injury in the brain.”
“The advances in the single-cell sequencing and low-input sequencing technologies we have developed together for capturing and processing the very sparse numbers of cells from these precious clinical samples were essential to collecting the data for this study,” adds J. Christopher Love, PhD, an associate member at the Ragon Institute and Raymond A. and Helen E. St. Laurent Professor in Chemical Engineering at the Koch Institute at MIT.
Britt Goods, PhD, co-first author on the study and a postdoctoral fellow working in Shalek’s lab, spent more than two years working on these rare samples, using RNA-sequencing techniques to profile specific immune cell populations. The RNA profiles of these cells provided insight into how the cells respond to stroke at different timepoints, and the researchers found some surprising results.
“Contrary to what we expected, we found that macrophages showed evidence of supporting a successful stroke response, driven primarily by certain metabolic factors,” says Goods.
Macrophages, Goods explains, are a type of immune cell that can drive harmful inflammation, and their complex role in the brain and central nervous system is often poorly understood in humans. In the samples from the stroke patients, they found that patients with good recovery had macrophages with activated glycolysis genes, which suggests a role for these metabolic pathways in promoting better outcomes.
“Even though this work is still preliminary, findings like this point to potential targets for future drug developments,” says Mike Askenase, PhD, co-first author of the study and an Associate Research Scientist in Sansing’s laboratory.
The RNA profiles of the cells can be used by other scientists to inform and drive research in in stroke and stroke recovery. This work, then, provides a valuable resource for the scientific community: a rare and detailed view of the cells hidden in the human brain.
Researchers at the Ragon Institute have developed a novel immunization strategy that shows promise for protecting against both group 1 and group 2 influenza A viruses. The strategy, detailed in their recent study, relies on eliciting a single amino acid change in antibodies to generate broadly neutralizing antibodies (bnAbs).
Their findings, to be published in Cell next month, reveal how the virus manipulates immune system processes to avoid destruction by natural killer (NK) cells, a type of white blood cell that is crucial for fighting viral infections.
The lab of the Ragon Institute faculty member Hernandez Moura Silva, PhD, recently published a review in Science Immunology regarding resident tissue macrophages (RTMs), shedding light on their multifaceted roles in organ health.