Date: March 25, 2013 By:
“Decades of research and three large-scale clinical trials have so far failed to yield an effective HIV vaccine, in large part because the virus evolves so rapidly that it can evade vaccine-induced immune responses.
Researchers from the Ragon Institute of Mass General, MIT, and Harvard University have now developed a new approach to vaccine design that may allow them to cut off those evolutionary escape routes. The researchers have developed and experimentally validated a computational method that can analyze viral protein sequences to determine how well different viral strains can reproduce in the body. That knowledge gives researchers an unprecedented guide for identifying viral vulnerabilities that could be exploited to design successful vaccine targets.
The team, led by Arup K. Chakraborty, the Robert T. Haslam Professor of Chemical Engineering, Chemistry, Physics and Biological Engineering at MIT, has designed protein fragments (peptides) that would target these weaknesses. Ragon Institute researchers are now developing ways to deliver the peptides so they can be tested in animals. ”
Read more at MIT News
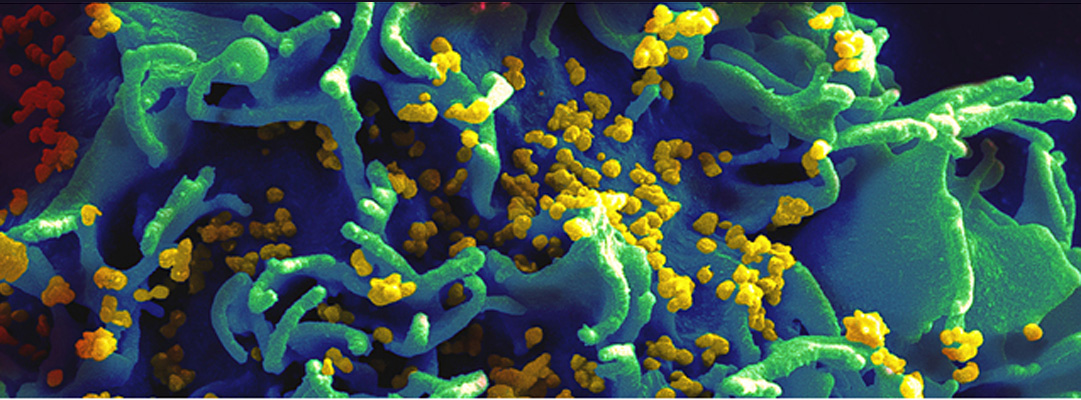
Image: This scanning electron micrograph shows HIV particles infecting a human T cell. (Source: National Institutes of Health)
Their findings, to be published in Cell next month, reveal how the virus manipulates immune system processes to avoid destruction by natural killer (NK) cells, a type of white blood cell that is crucial for fighting viral infections.
The lab of the Ragon Institute faculty member Hernandez Moura Silva, PhD, recently published a review in Science Immunology regarding resident tissue macrophages (RTMs), shedding light on their multifaceted roles in organ health.

After three years off due to the COVID-19 pandemic, the Ragon-MIT course HST.434 returned this January to provide 24 students a once in a lifetime learning experience