Date: April 27, 2022 By: Michael Morrison
BOSTON – More than 1.7 billion people—or a quarter of the world’s population—are infected with Mycobacterium tuberculosis, the bacterial strain that causes tuberculosis (TB). For years, scientists have been working to develop an effective vaccine, but current TB vaccines are only partially protective. New research by a team including investigators at Massachusetts General Hospital (MGH), Massachusetts Institute of Technology (MIT), the Ragon Institute of Mass General, MIT, and Harvard, and the Harvard T.H. Chan School of Public Health (Harvard Chan) has identified helpful and unhelpful aspects of the immune response that determine whether the body can keep TB infections under control. The findings, which are published in Immunity, may be helpful for designing a more effective vaccine.
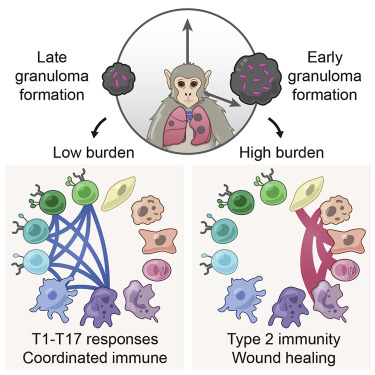
Although the body’s immune system can often keep M. tuberculosis infections under control so that people don’t experience any symptoms, there were more than 10 million active cases in 2017, with 1.6 million TB-related deaths. In response to active infection, which usually attacks the lungs, the body forms granulomas—tiny clusters of immune cells and other tissue-resident cells. In some granulomas, immune activity promotes bacterial clearance, but in others, bacteria persist and grow. These different granuloma responses can even be seen in the same individual.
“Identifying which cellular and molecular features associate with bacterial control could potentially point to new therapeutic and prophylactic strategies for TB,” says co–senior author Alex K. Shalek, PhD, a principal investigator who conducts research through the Ragon Institute of Mass General, MIT, and Harvard as well as through the Institute for Medical Engineering and Science, the Department of Chemistry, and the Koch Institute for Integrative Cancer Research at MIT and the Broad Institute of MIT and Harvard. To do so, Shalek and his colleagues used their single-cell profiling tools and teamed up with scientists in the lab of JoAnne Flynn, PhD, at the University of Pittsburgh School of Medicine, who had helped develop a monkey model that recapitulates the features of human TB and had also devised ways to track and quantify bacterial load and killing in individual granulomas.
“With TB, the immune response is pretty good but not great, and until recently, the field has tackled that problem with very rudimentary tools. This collaboration is bringing the very best tools and the very best minds to bear on a really hard, really important problem that most of the world ignores because it primarily impacts the global poor,” says co–senior author Sarah Fortune, MD, chair of the Department of Immunology and Infectious Diseases at Harvard Chan and an associate member of the Ragon Institute.
The collaborators’ efforts revealed that bacterial persistence occurs in granulomas enriched with certain cells—specifically mast, endothelial, fibroblast and plasma cells—that signal amongst themselves via particular pathways. Granulomas that supported bacterial clearance are characterized by other kinds of cells—including type 1-type 17, stem-like, and cytotoxic T cells—and use other types of signaling pathways.
“Our findings highlight new targets—such as specific cell subsets—to guide next-generation vaccines,” says Shalek. “We can also begin to consider how we might directly manipulate entire granulomas through modulating intercellular signaling to combat the bug more effectively.”
Fortune stresses that until the COVID-19 pandemic, TB was the leading cause of death from infectious disease worldwide. “An effective vaccine is the only way that we are really going to control TB, which shares many of the features that has made controlling COVID-19 hard: its airborne transmission, its infection in many people, and its ability to transmit before people are diagnosed,” she says. Unlike COVID-19, which is caused by a virus, TB is caused by bacteria, and current treatment involves multiple months of aggressive antibiotic therapy. “This study’s new insights into how the immune system clears, or in some cases helps, TB are critical in figuring out what a new vaccine should do,” says Fortune.
This work was supported by the Bill and Melinda Gates Foundation, Searle Scholars Program, the Beckman Young Investigator Program, Sloan Fellowship in Chemistry, the National Institutes of Health, the American Lung Association, the National Science Foundation, Fannie and John Hertz Foundation Fellowship, and Wellcome Trust Fellowship.
About the Massachusetts General Hospital
Massachusetts General Hospital, founded in 1811, is the original and largest teaching hospital of Harvard Medical School. The Mass General Research Institute conducts the largest hospital-based research program in the nation, with annual research operations of more than $1 billion and comprises more than 9,500 researchers working across more than 30 institutes, centers and departments. In August 2021, Mass General was named #5 in the U.S. News & World Report list of “America’s Best Hospitals.” MGH is a founding member of the Mass General Brigham health care system.
About the Ragon Institute
The Ragon Institute of Mass General, MIT, and Harvard was established in 2009 with a gift from the Phillip T. and Susan M. Ragon Foundation, with a collaborative scientific mission among these institutions to harness the immune system to combat and cure human diseases. Focusing on global infectious diseases, the Ragon Institute draws scientists, clinicians and engineers from diverse backgrounds and areas of expertise to study and understand the immune system with the goal of benefiting patients. For more information, visit www.ragoninstitute.org.
Their findings, to be published in Cell next month, reveal how the virus manipulates immune system processes to avoid destruction by natural killer (NK) cells, a type of white blood cell that is crucial for fighting viral infections.
The lab of the Ragon Institute faculty member Hernandez Moura Silva, PhD, recently published a review in Science Immunology regarding resident tissue macrophages (RTMs), shedding light on their multifaceted roles in organ health.

After three years off due to the COVID-19 pandemic, the Ragon-MIT course HST.434 returned this January to provide 24 students a once in a lifetime learning experience